Temperature is a concept that we use every day to describe how hot or cold something is. But what does temperature actually measure? And how is it related to the motions of molecules, the tiny particles that make up everything around us? In this article, we will explore these questions and learn some basic facts about temperature and molecular motion.
Contents
What is Temperature?
According to Chemistry LibreTexts, temperature is a measure of how hot or cold an object is relative to another object. It is also a measure of the average kinetic energy of the particles in matter. Kinetic energy is the energy of motion, and it depends on the mass and speed of the particle. The faster a particle moves, the more kinetic energy it has.
Temperature is not the same as heat, which is the flow of thermal energy between objects with different temperatures. Thermal energy is the total kinetic energy of all the particles in a system. When heat flows from a hotter object to a colder one, the thermal energy of the hotter object decreases and the thermal energy of the colder one increases. The temperature of both objects changes until they reach thermal equilibrium, which means they have the same temperature.
How are Temperature and Molecular Motion Related?
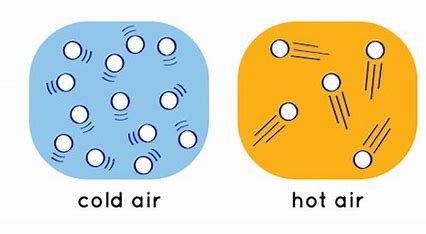
The motion of molecules in a system is related to its temperature by the average kinetic energy of the particles. The higher the temperature, the faster the particles move and the more often and harder they hit each other and the walls of their container. This causes the pressure of the system to increase. At absolute zero, which is -273.15°C or 0 K, the particles stop moving and have no kinetic energy.
Different states of matter have different degrees of molecular motion. In solids, the molecules are tightly packed and vibrate around fixed positions. In liquids, the molecules are more loosely packed and can slide past each other. In gases, the molecules are far apart and move freely in all directions. As matter changes from solid to liquid to gas, its temperature increases and so does its molecular motion.
How are Temperature Scales Defined?
There are three common scales used to measure temperature: Fahrenheit (°F), Celsius (°C), and Kelvin (K). Each scale has a different reference point and a different size of degree.
The Fahrenheit scale was developed by Daniel Gabriel Fahrenheit in 1724 and is based on arbitrary definitions of temperature. The freezing point of water is 32°F and the boiling point is 212°F. The Fahrenheit scale is typically not used for scientific purposes.
The Celsius scale was named after Anders Celsius in 1744 and is based on the properties of water. The freezing point of water is 0°C and the boiling point is 100°C. The Celsius scale is widely used in everyday life and in science.
The Kelvin scale was named after Lord Kelvin in 1848 and is based on absolute zero, which is the lowest possible temperature. The freezing point of water is 273.15 K and the boiling point is 373.15 K. The Kelvin scale is used for scientific measurements that require high precision and accuracy.
To convert between different temperature scales, we can use these formulas:
- °F = (°C × 1.8) + 32
- °C = (°F – 32) / 1.8
- K = °C + 273.15
- °C = K – 273.15
Summary
In this article, we learned that:
- Temperature is a measure of how hot or cold an object is relative to another object.
- Temperature is also a measure of the average kinetic energy of the particles in matter.
- Heat is the flow of thermal energy between objects with different temperatures.
- The motion of molecules in a system is related to its temperature by the average kinetic energy of the particles.
- Different states of matter have different degrees of molecular motion.
- There are three common scales used to measure temperature: Fahrenheit, Celsius, and Kelvin.
- Each scale has a different reference point and a different size of degree.
- We can convert between different temperature scales using simple formulas.